Recherche
ION CHANNELS AS DRUG TARGETS - Ions cannot move freely across cell membranes, but must enter or exit a cell through dedicated pores created by ion channels. This selective transport of ions through biological membranes underpins the most fundamental physiological processes. Ion channel activity, beside making the brain work, is necessary for fluid secretion in the lung, gastro-intestinal tract and kidney, muscular contraction, vascular tone, electrical signalling in the heart and the nervous system, as well as hormone secretion, immune response, and cell proliferation, amongst the most important functions. The biological significance of ion channels is underlined by their involvement in a wide variety of pathologies spanning all the major areas of therapeutic interest. Numerous diseases associated with mutations in genes encoding ion channels have now been identified that affect cardiac, neuronal, neuromuscular, neurosensory, metabolic, epithelial and respiratory systems. These diseases, known as “channelopathies”, are not only inherited but can also be acquired such as autoimmunity directed against ion channels in both peripheral and central nervous systems.
Given their very diverse biological roles, it is not surprising that ion channels are major drug targets (1-3). Roughly 15% of known drugs have their primary action on ion channels, making them the second largest target class after G-protein coupled receptors. It is estimated that worldwide sales of ion channel modulators generate over 12 billion € in revenues each year. These modulators comprise drugs for local and general anesthesia, and for treating diabetes, hypertension, cardiac arrhythmia, pain, insomnia, anxiety and epilepsy. Examples of currently marketed drugs include calcium channel blockers, such as Norvasc and Verapamil, which are used for the treatment of hypertension and various other cardiovascular disorders; sodium channel blockers, such as Lidocaine, a local anesthetic, and Lamictal, used for the treatment of epilepsy; and potassium channel blockers, such as Glipizide, which is used in the treatment of diabetes. Despite being a promising source of new targets for a number of other pathologies, ion channels have proven to be particularly resistant to molecular-driven approaches to drug discovery. Almost all the current ion channel modulators were discovered using traditional animal-based pharmacological approaches, in many cases without knowledge of their molecular target. With the advent of a better understanding of cellular physiology and identification of the molecular components that constitute individual channel types and/or control their activity, rational molecular-based strategies to identify ion channel modulators are now within reach. Such approaches have been strengthened by the advent of new technologies including high throughput screening (HTS) assays and automated electrophysiology.
ION CHANNEL COMPLEXITY AND THERAPEUTIC OPPORTUNITIES - Ion channels are multimeric protein assemblies comprising core subunits forming the ionic pore and accessory subunits. More than 350 human genes encode pore-forming subunits. Alternative splicing, editing and heteromultimerization of pore-forming subunits are mechanisms that further increase this diversity. There are no tissue-specific ion channels but complex expression patterns of multiple ion channel subunits in each cell type. This huge structural diversity is a major challenge for molecular drug discovery. In many cases, the exact subunit composition of a target channel in the tissue of interest is only poorly characterized. Very often, the functional roles of the individual channel types are not well defined. Another challenge is the very dynamic behavior of ion channels that ultimately determines the macroscopic currents at the plasma membrane. The different steps of their biogenesis, addressing, recycling and degradation are shown in Figure 1. These different steps require cellular factors that are only poorly understood.
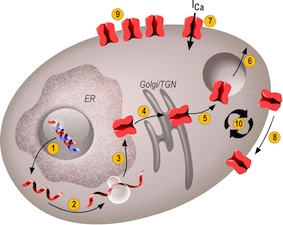
Figure 1. Macroscopic currents at the plasma membrane depend on a dynamic set of regulated steps. Biogenesis steps include gene translation and splicing/editing (1), ribosomal translation (2), folding and assembly in the endoplasmic reticulum (ER) (3), export from the ER to the Golgi apparatus for maturation (4), then budding from the trans-Golgi network (TGN) and sorting (5). Once at the plasma membrane, besides direct regulations affecting channel activity (7), ion channels may be addressed to specific membrane signaling domains (8), clustered (9) and recycled/degraded (10).
Identification of novel targets requires a thorough understanding of the processes driving channel diversity and regulation. Their validation as relevant targets requires testing the physiological relevance of the identified mechanisms by functional exploration in cell and animal models where ion channels and regulatory cellular factors can be manipulated. The goal is not only to validate active channels at the plasma membrane as targets for drug discovery, but also mechanisms controlling ion channel dynamics that are therapeutic opportunities for the next generation of channel modulators.
ICST OBJECTIVES - The global goal is to form a strong network sharing expertise, tools and methods in the area of ion channels to create and/or enhance synergies among the various groups. The benefit will be a more global knowledge about ion channels, from genes to molecules to humans, that will result in identification and validation of new targets for drug discovery. The most interesting results will be patented and licensed to private companies including biotech spinoffs.
The quality of the fundamental research carried by the consortium groups and the diversity of their scientific questions is a strength and a unique opportunity. Rather than diverting research teams from their current objectives by immediately putting emphasis on a few focused scientific questions, the research program aims to identify a few general axes to develop that can really bring a plus to the consortium by increasing synergy between the teams. The benefit of this approach should lead each team to adapt its scientific objectives progressively during the next years and will allow accomodation of new teams along the program. Two axes are related to the identification and validation of new drug targets. By adopting complementary technical approaches and by sharing results, each team will be able to expand its own interests to other channel types and pathologies. At the fundamental level, we expect to identify general mechanisms of regulation that hold for different types of ion channels (for example in trafficking and recycling). The 3rd axis is related to new methodological approaches to study ion channels that will benefit the whole consortium. A private company, Cisbio Bioassays will join the consortium for adapting its assays developed for G-protein coupled receptors to ion channels. The consortium will provide its expertise concerning ion channel study and Cisbio bioassays will design new non-electrophysiological assays dedicated to ion channel study. The last axis is related to the emergence of new teams and skills in the consortium, including zebrafish genetics and high resolution optical techniques and optogenetics. Intensive drug screening is clearly out the scope of ICST given the much better positioning of industry for this particular task.
Conception-Realisation Laure Tauc - InformaNice.